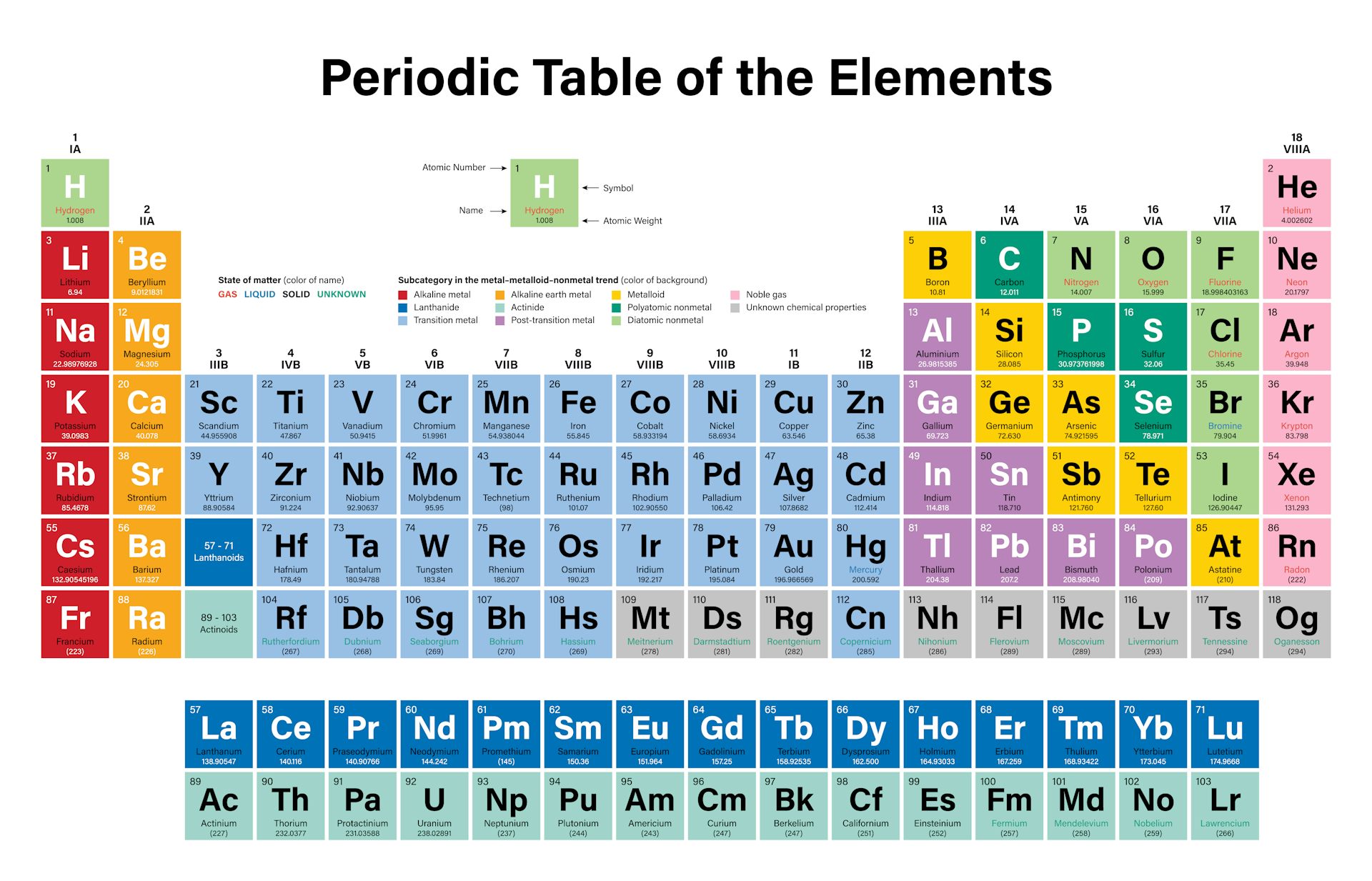
The most reactive metal is francium, the last alkali metal (and most expensive element ). Reactivity Trend in the Periodic Table The most reactive element is fluorine, the first element in the halogen group. Alloys, such as brass and bronze, also are metals. Most of the elements on the periodic table are metals, including gold, silver, platinum, mercury, uranium, aluminum, sodium, and calcium. What are the basic metals on the periodic table? The two most reactive would be Fluorine and (theoretically) Francium. Those groups have 1 and 7 valence electrons, respectively, making them desperate to bond to something, to achieve a stable configuration of 8. The two most reactive groups of elements are the alkali metals and the halogens, because of their valence electrons. What groups are the most reactive metals? Highly reactive metals (potassium, sodium) are placed at the top of the reactivity series, whereas least reactive metals (silver, gold) are placed at the bottom of the reactivity series. Which is the most reactive metals Class 8? Fluorine gas reacts explosively with many other elements and compounds and is considered to be one of the most dangerous known substances. It is not found in nature as a free element. The element fluorine is the most reactive nonmetal. Which is the most reactive metal and non metal? The most reactive metals in the periodic tables are the alkali metals, followed by the alkaline earth metals. What are the most reactive families on the periodic table? Platinum is the least reactive of all the options. Which is the least reactive metal in the periodic table? Cesium is second from the bottom of this group, with 6 electron shells, so it fits all the characteristics of a reactive atom, therefore making it the most reactive element. What is the most reactive element and why?Īlkali metals (situated far away from transitional metals and noble gases) are the most reactive elemental group. In a reactivity series, the most reactive element is placed at the top and the least reactive element at the bottom….The reactivity series. As you move down the column, the metals become more reactive because the nucleus gains more electrons and protons (more electron levels), weakening their electrostatic force. Caesium is the most reactive metal in the periodic table, so much that working with this metal often ends in explosions!Įxplanation: Alkali metals are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). What is the most reactive on the periodic table?įluorine is identified as the most reactive nonmetal and the most electronegative element in the periodic table, making it the strongest oxidizing agent. Francium, however, is a laboratory-produced element and only minute quantities have been made, so for all practical purposes, the most reactive metal is cesium. The most reactive metal on the periodic table is francium. What is the most reactive metal in period? Lithium, sodium, and potassium all react with water, for example. 
The elements toward the bottom left corner of the periodic table are the metals that are the most active in the sense of being the most reactive. 
Where are the most reactive metals on the periodic table? 
9 What is the most reactive metal group?.8 What are the basic metals on the periodic table?.7 What groups are the most reactive metals?.6 What are the most reactive families on the periodic table?.5 Which is the least reactive metal in the periodic table?.4 What is the most reactive element and why?.3 What is the most reactive on the periodic table?.2 What is the most reactive metal in period?.1 Where are the most reactive metals on the periodic table?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
